ABOUT US
- HOME
- ABOUT US
WHO WE ARE
A Few Words Of Our COMPANY
Bright Pharma Engineering is a leading and well-known processing equipment manufacturer for the pharmaceutical, bio-pharma, healthcare, and cosmetic industries. Our strength lies in our team of dedicated and trained engineers and technicians with over 15 to 20 years of experience. We serve the needs of more than 1,000 customers both in India and abroad, with a reputation for providing quality service, on-time delivery, and user-friendly designs.
- Established In 1995
- Manufacturing Facilities at Vasai & Vadodora
- Factory Acceptance Testing (F.A.T.) Facility
- Manufacturing & Integration

WHO WE ARE
A Few Words Of Our COMPANY

Bright Pharma Engineering is a leading and well-known manufacturer of processing equipment for the pharmaceutical, bio-pharma, healthcare, and cosmetic industries. Our strength lies in our team of dedicated and trained engineers and technicians, who have over 15 to 20 years of experience. We serve the needs of more than 1,000 customers both in India and abroad, with a reputation for providing quality service, on-time delivery, and user-friendly designs.
- Established In 1995
- Manufacturing Facilities At Vasai & Vadodora
- Factory Acceptance Testing (F.A.T.) Facility
- Manufacturing & Integration
- Established In 1995
- Manufacturing Facilities At Vasai & Vadodora
- Factory Acceptance Testing (F.A.T.) Facility
- Manufacturing & Integration
Manufacturing Infrastructure
We have state-of-the-art facilities covering an area of over 25,000 sqft., in Vasai, Maharashtra, and 56,000 sqft., in Vadodara, Gujarat, India. Our plants are fully integrated with fabrication, assembly, and testing facilities, all managed by skilled technicians and engineers. A dedicated area for Factory Acceptance Testing (FAT) allows us to test all our systems under various process conditions before they are shipped to the site. Our equipment consistently performs well, is easy to operate, and is designed to be compact and reliable with low operational life-cycle costs. They are engineered and manufactured in compliance with international standards and guidelines, such as US FDA and EU GMP.
Manufacturing Infrastructure

We have state-of-the-art facilities covering an area of over 25,000 sqft., in Vasai, Maharashtra, and 56,000 sqft., in Vadodara, Gujarat, India. Our plants are fully integrated with fabrication, assembly, and testing facilities, all managed by skilled technicians and engineers. A dedicated area for Factory Acceptance Testing (FAT) allows us to test all our systems under various process conditions before they are shipped to the site.
Our equipment consistently performs well, is easy to operate, and is designed to be compact and reliable with low operational life-cycle costs. They are engineered and manufactured in compliance with international standards and guidelines, such as US FDA and EU GMP.
Our TEAM



WHAT WE OFFER
Manufacturing WORKFLOW
Manufacturing is a straightforward process in which raw materials or parts are purchased, assembled, and then transformed into a finished product, known as the manufacturing workflow. A manufacturing workflow consists of a series of processes that guide the work of equipment and personnel from start to finish. It can be broken down into five main steps:
- Planning
- Scheduling
- Executing
- Monitoring
- Controlling

1.
- P & 1D
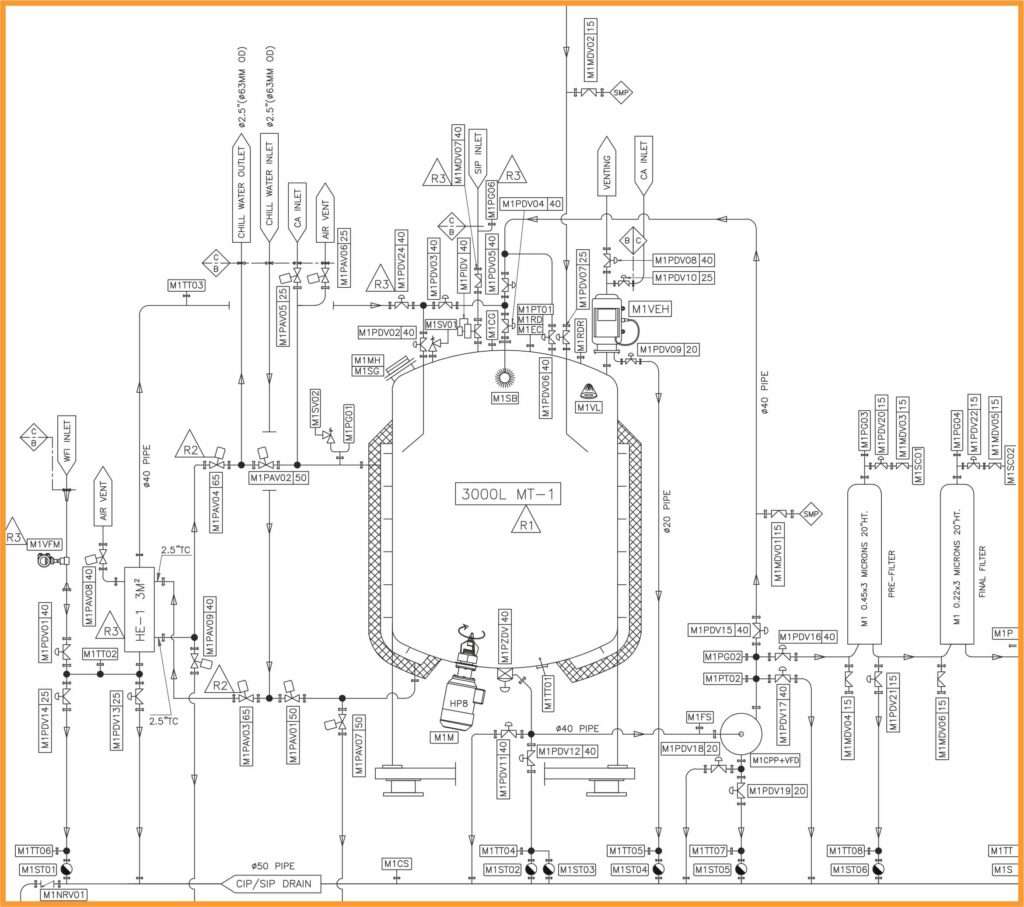
The manufacturing process begins with the preparation and approval of P&ID drawings, which detail the arrangement of instruments, equipment, and components like vessels, pumps, and valves. Upon approval, the next step is creating the General Assembly (GA) drawings.
2.
- General Assembly Drawing
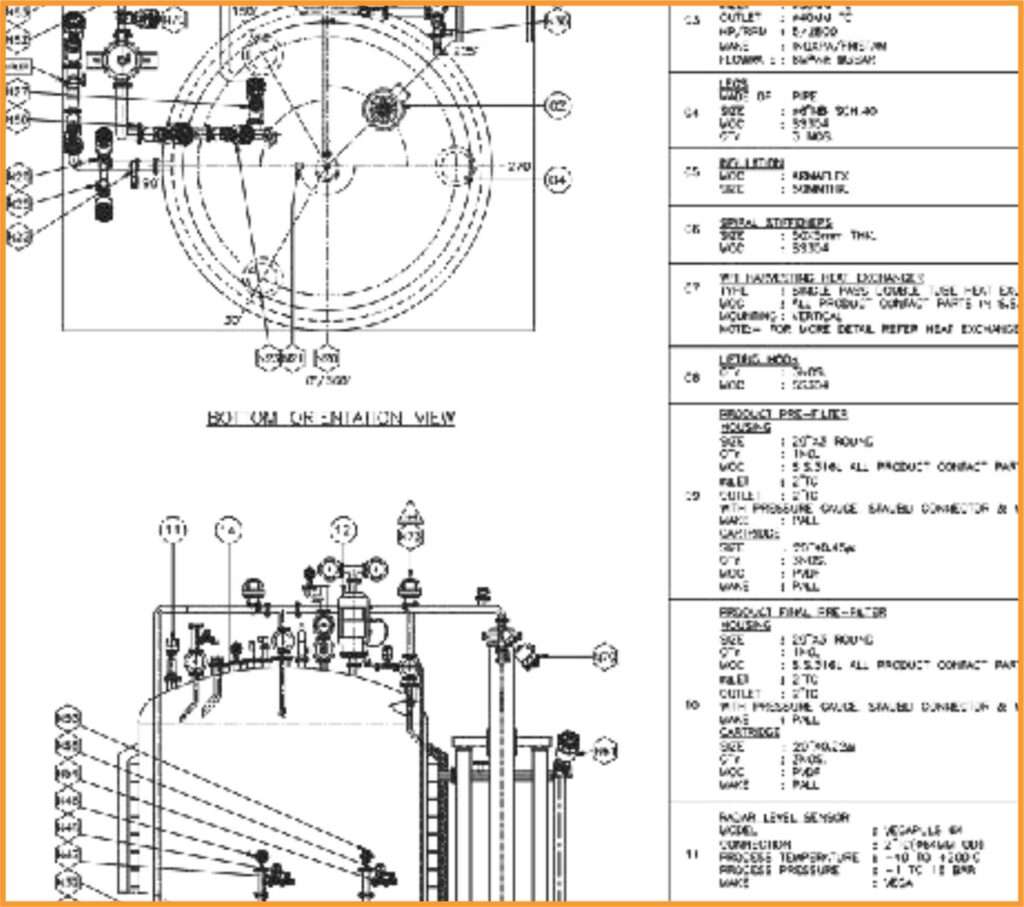
The General Assembly (GA) drawings provide details such as dimensions, design parameters, surface finish quality (Ra values), and BO specifications. Once approved, we move to the next stage: 3D system design.
3.
- 3D Model Preparation

Using cutting-edge 3D visualization and simulation software for engineering and design purposes. These advanced tools help us understand integration, size, scale, and potential issues during fabrication, installation, and system integration.
4.
- System Execution

The systems are built, put together, and tested with the aid of the 3D model. Delays are reduced by the 3D software’s efficient assembly and integration processes. Following assembly, the system is examined to make sure there are no deviations from the P&ID, GA, and 3D models.
- SYSTEM ENGINEERING
- SYSTEM MANUFACTURING
- QUALITY CONTROL SYSTEMS
- INFRASTRUCTRE
- PROJECT EXPERTISE
- TECHNICAL SUPPORT
- VALIDATION SUPPORT
- SERVICE SUPPORT
WHAT WE OFFER
Manufacturing WORKFLOW

Manufacturing is a straightforward process in which raw materials or parts are purchased, assembled, and then transformed into a finished product, known as the manufacturing workflow. A manufacturing workflow consists of a series of processes that guide the work of equipment and personnel from start to finish. It can be broken down into five main steps:
- Planning
- Scheduling
- Executing
- Monitoring
- Controlling
1.
- P & 1D

The manufacturing process begins with the preparation and approval of P&ID drawings, which detail the arrangement of instruments, equipment, and components like vessels, pumps, and valves. Upon approval, the next step is creating the General Assembly (GA) drawings.
2.
- General Assembly Drawing

The General Assembly (GA) drawings provide details such as dimensions, design parameters, surface finish quality (Ra values), and BO specifications. Once approved, we move to the next stage: 3D system design.
3.
- 3D Model Preparation

Using cutting-edge 3D visualization and simulation software for engineering and design purposes. These advanced tools help us understand integration, size, scale, and potential issues during fabrication, installation, and system integration.
4.
- System Execution

The systems are built, put together, and tested with the aid of the 3D model. Delays are reduced by the 3D software’s efficient assembly and integration processes. Following assembly, the system is examined to make sure there are no deviations from the P&ID, GA, and 3D models.
- SYSTEM ENGINEERING
- SYSTEM MANUFACTURING
- QUALITY CONTROL SYSTEMS
- INFRASTRUCTRE
- PROJECT EXPERTISE
- TECHNICAL SUPPORT
- VALIDATION SUPPORT
- SERVICE SUPPORT
- SYSTEM ENGINEERING
- SYSTEM MANUFACTURING
- QUALITY CONTROL SYSTEMS
- INFRASTRUCTRE
- PROJECT EXPERTISE
- TECHNICAL SUPPORT
- VALIDATION SUPPORT
- SERVICE SUPPORT
WHY CHOOSE US
Our ADVANTAGES
Our distinctiveness comes from our proficiency in manufacturing machines for Pharmaceutical, Cosmetic, and Bio-Products. With a thorough awareness of the demands unique to each industry, we provide customized solutions that guarantee excellent efficiency, compliance, and performance for various applications.
Understanding that every business has unique needs, we offer customized machine solutions. Our expert engineers work closely with you to design and manufacture equipment that perfectly aligns with your operational requirements, ensuring maximum efficiency and productivity.
The core of our production process is quality. We ensure that every equipment we make satisfies the highest requirements of durability and dependability by putting in place strict quality control systems and testing protocols. This gives you piece of mind.
Our dedication to client satisfaction goes beyond the point of delivery. We provide committed support services, guaranteeing prompt help and thorough fixes for any problems you could run into. Our responsive team is constantly available to assist, ensuring continuous operations.
MISSION
Focused on quality, supplying top-notch process equipment for various industries.
VISSION
Emphasizing learning, interaction, and innovative work ethics.
VALIDATION
USFDA validation ensures consistent production meeting predetermined specifications and quality attributes.
Client TESTIMONIALS



